SUMMARY
A novel mechanism for 5-azacytidine-mediated depletion of RNA 5-methylcytosine on chromatin-associated RNA leading to global transcriptional repression through two epigenetic pathways enabling more precise and effective leukemia treatments
The Unmet Need: Optimized 5-azacytidine therapeutic strategies and reliable biomarkers
-
Epigenetic regulation, which involves chemical modifications to DNA and RNA that affect gene expression without altering the genetic code, has emerged as a critical field in understanding and treating cancers such as myelodysplastic syndromes (MDS) and acute myeloid leukemia (AML). Traditional therapies have focused on targeting DNA methylation, a process where methyl groups are added to DNA, often silencing tumor suppressor genes and contributing to malignancy. Drugs that inhibit DNA methylation, such as 5-azacytidine (5-azaC) and decitabine, have been widely used in clinical practice to reverse these epigenetic changes and restore normal gene function. However, despite their broad application, patient responses are highly variable, and many individuals either fail to respond or eventually develop resistance. This variability underscores a pressing need for a deeper understanding of the molecular mechanisms underlying drug action and resistance, as well as for the development of more precise biomarkers to guide therapy.
-
Current approaches to epigenetic therapy, particularly those targeting DNA methylation, face significant limitations. The prevailing assumption has been that the therapeutic effects of agents like 5-azaC and decitabine derive primarily from their ability to demethylate DNA and reactivate silenced genes. However, these drugs often show delayed or incomplete responses, and their effects on global gene expression are not always consistent with DNA demethylation alone. Furthermore, the toxicity associated with non-specific DNA demethylation can limit dosing and reduce therapeutic windows, while resistance mechanisms—such as mutations in DNA methyltransferases or other epigenetic regulators—can further compromise outcomes. These challenges highlight the urgent need for alternative strategies that address the shortcomings of current DNA-centric approaches and provide a more nuanced understanding of epigenetic regulation in cancer.
The Proposed Solution: Novel method using 5-azacytidine to deplete RNA methylation leading to gene repression enabling more precise and effective leukemia treatment
-
The faculty inventor uncovered a novel mechanism of action for 5-azacytidine, a drug used in the treatment of myeloid malignancies such as myelodysplastic syndromes (MDS) and acute myeloid leukemia (AML). Traditionally, 5-azaC was thought to work primarily by causing DNA hypomethylation, but the inventor reveals that its main cytotoxic effect is mediated through the rapid depletion of 5-methylcytosine (m5C) on chromatin-associated RNA (caRNA). This RNA modification loss leads to global transcriptional repression suppressing genes critical for cell cycle progression and DNA repair, ultimately inducing leukemia and cell death.
FIGURE
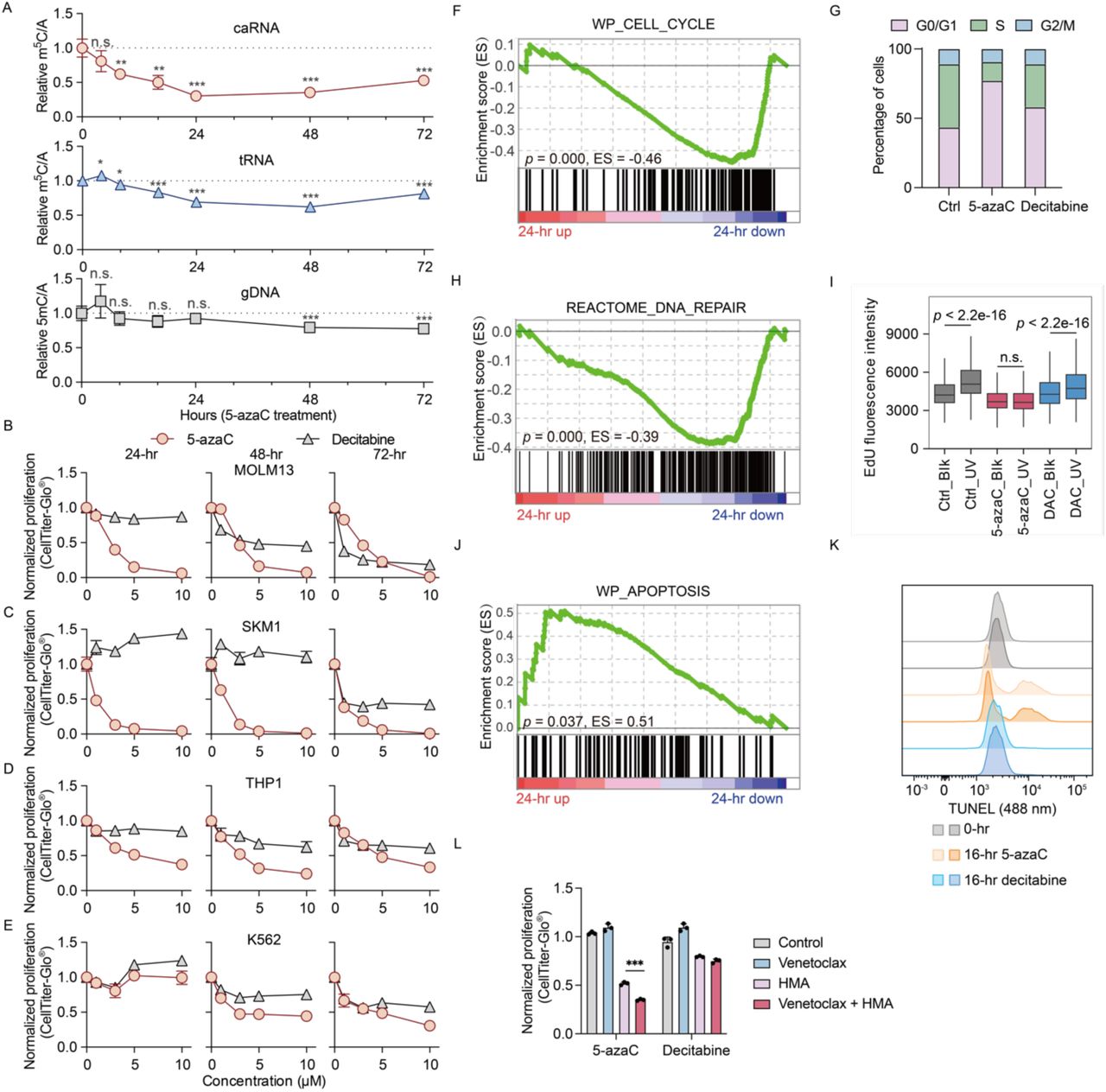
5-azaC induces RNA m5C depletion and leukemia cell death at early time points.
ADVANTAGES
ADVANTAGES
-
Targets RNA 5-methylcytosine (m5C) depletion to induce leukemia cell death, providing a novel mechanism distinct from DNA hypomethylation
-
Induces global transcriptional repression of genes critical for cell cycle and DNA repair, enhancing cytotoxic effects on leukemia cells
-
Identifies key RNA methyltransferase targets, enabling precise disruption of RNA methylation
-
Provides biomarkers for predicting patient sensitivity and personalizing 5-azaC therapy
-
Suggests potential combination therapy targets to enhance treatment efficacy and overcome resistance
APPLICATIONS
-
Personalized leukemia treatment stratification
-
Development of RNA methylation inhibitors
-
Combination therapies targeting epigenetic pathways
-
Companion diagnostics for 5-azaC response
-
Biomarker-driven clinical trial design
PUBLICATIONS
-
5-azacytosine induces cytotoxicity via 5-methylcytosine depletion on chromatin-associated RNA in leukemia. Boyang Gao, Ying Li, Long Zhao, Zhongyu Zou, Xinyuan Ma, Juyeong Hong, Junhong Xiang, Xiaoyang Dou, Feng-Chun Yang, Mingjiang Xu, Chuan He. bioRxiv 2025.06.24.661348; doi: https://doi.org/10.1101/2025.06.24.661348
October 29, 2025
Proof of concept
Patent Pending
Licensing,Co-development
Chuan He