SUMMARY
Advanced imaging and targeted radiotherapy for neuroendocrine tumors, using DOTATATE-based agents for precise diagnosis and treatment, and enhancing therapy effectiveness by adding Fulvestrant as a radiosensitizer to further reduce tumor growth
The Unmet Need: More effective neuroendocrine tumor treatments that can further improve patient outcomes
- Neuroendocrine tumors (NETs) represent a diverse and increasingly diagnosed group of malignancies that originate from neuroendocrine cells and frequently express somatostatin receptors, particularly SSTR2. The advent of radiolabeled somatostatin analogs (RSAs) has revolutionized the diagnosis and management of NETs, allowing for highly sensitive and specific imaging as well as targeted radionuclide therapy. Diagnostic tools such as 68Ga-DOTATATE PET/CT have become the gold standard, offering superior image quality and lower radiation exposure compared to older modalities. For treatment, peptide receptor radionuclide therapy (PRRT) with agents like 177Lu-DOTATATE has provided a much-needed option for patients with advanced, unresectable, or progressive NETs, especially after standard somatostatin analog therapy has failed.
-
While PRRT with 177Lu-DOTATATE has demonstrated significant benefits in terms of progression-free survival and symptom control, several limitations persist with current approaches. Not all patients derive the same degree of benefit, with therapeutic efficacy closely tied to the level of somatostatin receptor expression, tumor burden, and lesion size. Objective tumor response rates, while promising, still leave a substantial proportion of patients with residual or progressive disease. Additionally, the overall survival advantage remains less definitive, and the risk of adverse effects such as renal toxicity and hematologic complications necessitates careful patient selection and monitoring. Efforts to enhance PRRT efficacy—such as combining different radionuclides, utilizing alpha emitters, or pairing PRRT with other systemic therapies—have yet to yield a universally accepted solution. As such, there remains a critical need for innovative strategies that can sensitize tumors to radiotherapy and overcome the current therapeutic plateau.
The Proposed Solution: Novel therapeutic approach combines Fulvestrant as a radiosensitizer with 177-Lu-DOTATATE (Lutathera) for neuroendocrine tumor treatment
- The faculty inventor developed an advanced approach to the diagnosis and treatment of neuroendocrine tumors (NETs) by leveraging radiolabeled somatostatin analogs (RSAs), particularly DOTATATE-based compounds, in both imaging and therapy. For diagnosis, agents like 68Ga-DOTATATE and 64Cu-DOTATATE enable highly sensitive and specific PET/CT imaging of NETs, facilitating accurate staging, surgical planning, and monitoring of disease progression. For treatment, 177Lu-DOTATATE (Lutathera) is used in peptide receptor radionuclide therapy (PRRT) for patients with advanced, unresectable, or progressive NETs that express somatostatin receptors. The standard protocol involves multiple cycles of 177Lu-DOTATATE, with concurrent amino acid infusions to reduce renal toxicity. This comprehensive approach not only improves disease stabilization and symptom control but also allows for tailored patient selection based on somatostatin receptor imaging.
-
What differentiates this solution is the integration of Fulvestrant, a radiosensitizing agent, with 177Lu-DOTATATE therapy. The use of Fulvestrant represents a novel strategy to overcome resistance and improve outcomes, building on the established success of DOTATATE-based imaging and therapy. This innovation positions the technology at the forefront of NET management, offering a promising path to more effective and personalized treatment regimens.
FIGURE
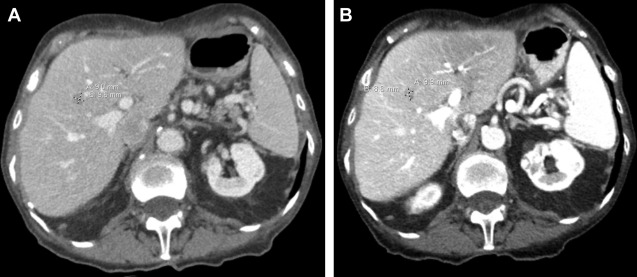
Surveillance CT scans for a patient with small bowel neuroendocrine tumor metastatic to the liver demonstrating stable/nonresponsive disease to peptide receptor radionuclide therapy (PRRT) during the interval following (A) PRRT cycle #2 to (B) completion of PRRT cycle #4.
ADVANTAGES
ADVANTAGES
-
Enhanced therapeutic efficacy of 177Lu-DOTATATE through radiosensitization by Fulvestrant, leading to significantly reduced tumor growth
-
Improved treatment outcomes for neuroendocrine tumors, especially metastatic and unresectable cases
-
Targeted therapy leveraging somatostatin receptor expression for precise tumor localization and treatment
-
Potential for better progression-free survival and quality of life compared to standard therapies
-
Combination therapy may allow for lower doses or fewer cycles of radiotherapy, potentially reducing side effects
APPLICATIONS
-
Advanced neuroendocrine tumor therapy
-
Enhanced PRRT for metastatic NETs
-
Combination radiosensitizer clinical trials
PUBLICATIONS
May 30, 2024
Proof of concept
Patent Pending
Licensing,Co-development
Xavier Keutgen
- Pre-clinical studies have shown that Fulvestrant significantly enhances the tumor-killing effects of 177Lu-DOTATATE, resulting in greater tumor reduction compared to PRRT alone