SUMMARY
Detection and monitoring of autoantibodies against Angiotensin II in COVID-19 patients, guiding diagnosis, treatment, and vaccine design to reduce blood pressure complications and prevent harmful autoimmune responses linked to the virus’s Spike protein
The Unmet Need: Novel strategies to understand and mitigate the unpredictable cardiovascular and respiratory deterioration observed in viral infection hospitalized patients
- The management of severe viral infections, particularly SARS-CoV-2, represents a critical intersection between infectious disease immunology and cardiovascular health. While the immune system mounts a defense against viral antigens, this response can become dysregulated, leading to severe complications. Specifically, there is an urgent requirement to identify the biological mechanisms causing sudden blood pressure fluctuations and severe hypotension to improve patient triage and prevent long-term vascular damage.
-
Current clinical approaches fail to account for epitope mimicry, where the immune response to a virus inadvertently attacks the host's own regulatory proteins. Existing diagnostic protocols do not screen for autoantibodies targeting vasoconstrictive peptides, leaving clinicians blind to a major cause of vascular instability. Furthermore, standard therapeutic antibodies are not routinely evaluated for cross-reactivity with host cardiovascular regulators, risking the administration of harmful autoantibodies. Similarly, conventional vaccine design focuses on maximizing viral neutralization without adequately screening for autoimmune induction. Consequently, current immunizations and therapies can inadvertently trigger severe blood pressure dysregulation, highlighting a critical flaw in intervention development.
The Proposed Solution: Platform to detect Angiotensin II autoantibodies to optimize treatment plans for infected and vaccinated individuals
- The faculty inventor developed methods to address COVID 19-induced autoimmunity by detecting and mitigating autoantibodies that target Angiotensin II (AngII). The technology features diagnostic assays, such as ELISA and lateral flow tests, to quantify anti-AngII levels in patient blood, serving as a biomarker for blood pressure dysregulation and respiratory distress. Additionally, the platform provides screening protocols to ensure therapeutic monoclonal antibodies neutralize the virus without cross-reacting with host AngII. It also encompasses advanced vaccine design strategies, utilizing targeted protein engineering to mutate specific Spike protein sequences and optimizing adjuvant selection to prevent the induction of harmful autoimmune responses.
Figure
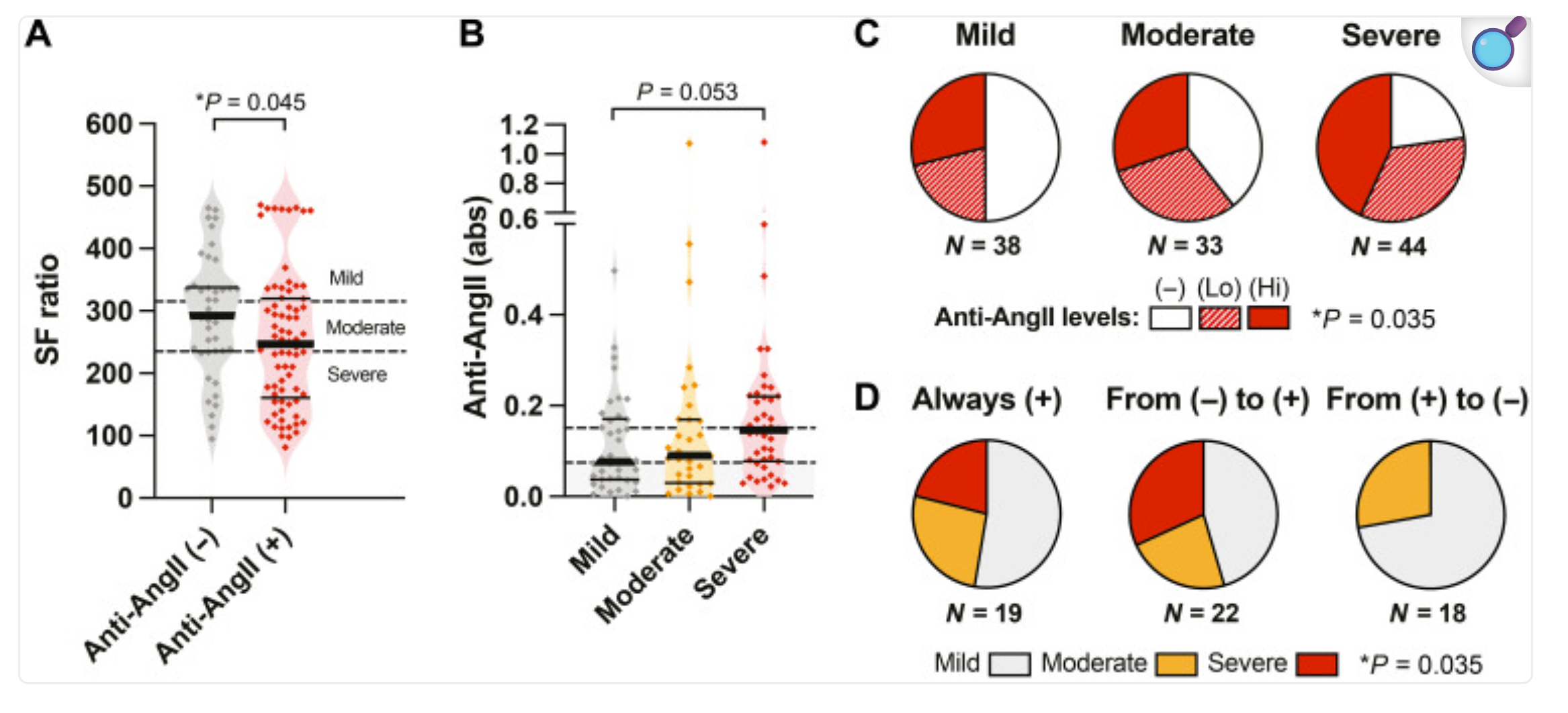
Anti-AngII autoantibodies correlates with reduced pulse oxymetric saturation SpO2/FiO2 (SF ratio) in hospitalized patients with COVID N = 115)
ADVANTAGES
ADVANTAGES
- Provides a novel biomarker to accurately predict disease severity, respiratory distress, and blood pressure dysregulation in patients with a history of coronavirus infection
-
Enables early and targeted clinical interventions, such as the timely administration of vasopressors, by identifying patients at high risk for acute hypotension
-
Improves the safety of therapeutic monoclonal antibodies and inhibitors by providing a screening method to eliminate candidates that cross-react with host Angiotensin II
-
Guides the development of safer vaccines through targeted protein engineering and optimal adjuvant selection, minimizing the risk of triggering autoimmune responses
-
Helps prevent adverse vascular events and blood pressure instability during both viral treatment and immunization
APPLICATIONS
-
COVID-19 severity diagnostic kits
-
Blood pressure dysregulation screening
-
Therapeutic antibody safety screening
-
Safer COVID-19 vaccine development
-
Clinical trial safety monitoring
PUBLICATIONS
- Briquez PS, Rouhani SJ, Yu J, Pyzer AR, Trujillo J, Dugan HL, Stamper CT, Changrob S, Sperling AI, Wilson PC, Gajewski TF, Hubbell JA, Swartz MA. Severe COVID-19 induces autoantibodies against angiotensin II that correlate with blood pressure dysregulation and disease severity. Sci Adv. 2022 Oct 7;8(40):eabn3777. doi: 10.1126/sciadv.abn3777. Epub 2022 Oct 7. PMID: 36206332; PMCID: PMC9544317
May 30, 2024
Proof of concept
Patent Pending
Licensing,Co-development
Melody Swartz