SUMMARY
A method to attenuate RSV and other NNS RNA viruses by reducing m6A modifications on their RNA. This approach enhances genetic stability, ensures sufficient attenuation, triggers higher immune responses, retains high immunogenicity, and makes vaccine production feasible.
The problem: A live attenuated vaccine is one of the most promising vaccine approaches for RSV. However, a major problem is that natural RSV infection does not induce long-lived protection.
- Non-segmented negative-sense (NNS) RNA viruses encompass a wide range of significant human, animal, and plant pathogens in families such as Paramyxoviridae, Pneumoviridae, Rhabdoviridae, Filoviridae, and Bornaviridae. These viruses, including human respiratory syncytial virus (RSV), pose substantial health risks, particularly for infants, young children, the elderly, and immunocompromised individuals. For example, RSV is responsible for significant respiratory infections and has a high hospitalization and mortality rate in children under five years of age.
-
Despite extensive research efforts, effective vaccines or antiviral treatments against RSV and other NNS RNA viruses remain unavailable. Given the global health impact, there is a critical need for new approaches to develop effective vaccines for these pathogens.
-
Current methods to attenuate live viruses for vaccine use have several limitations. Traditional approaches often involve gene deletions or mutations achieved under stress conditions, but these methods can be simplistic and might not provide lasting attenuation due to the risk of reversion to a more virulent form. Another approach involves modifying structural proteins which can occasionally hinder the vaccine efficacy. Furthermore, traditional attenuation techniques might not fully prevent the virus from retaining pathogenic qualities.
-
These limitations underscore the necessity for novel methodologies that ensure genetic stability and sufficient attenuation without compromising immunogenicity. Addressing these issues is essential to providing safer and more effective live attenuated vaccines against RSV and other NNS RNA viruses.
The proposed solution: A novel strategy employing recombinant RSVs deficient in viral RNA (m6A) to enhance innate immune response which in turn promotes an adaptive immune response to RSV infection
- The faculty inventor, Chaun He, developed a method for the attenuation of non-segmented negative-sense (NNS) RNA viruses, specifically respiratory syncytial virus (RSV), by targeting the N6-methyladenosine (m6A) modifications in their RNA. Normally, m6A modifications enhance viral replication and pathogenesis. The technology employs a recombinant RSV that lacks m6A sites in the G gene transcript, leading to defective replication and reduced pathogenicity in models like A549 cells and cotton rats' respiratory tracts.
-
Notably, m6A-deficient RSV, such as rgRSV-G12, is significantly attenuated but retains high immunogenicity, offering complete protection against RSV. Thus, viral m6A methylation serves as a novel target for designing live attenuated vaccines for RSV and potentially other NNS RNA viruses.
-
This approach is differentiated due to its multi-faceted genetic modifications that enhance vaccine strain stability and reduce the risk of reversion to a wild-type virus. Unlike traditional methods involving single mutations, multiple modifications in m6A sites mean reversion requires numerous genetic reversals, making it highly unlikely. Furthermore, m6A-deficient viruses maintain high immunogenicity and can elicit stronger innate immune responses, enhancing vaccine efficacy.
-
The potential for applying this strategy across diverse NNS RNA viruses, combined with its ability to produce viruses at variable attenuation levels and economically feasible titers, underscores its universal applicability and superiority over existing vaccine generation methods.
FIGURE
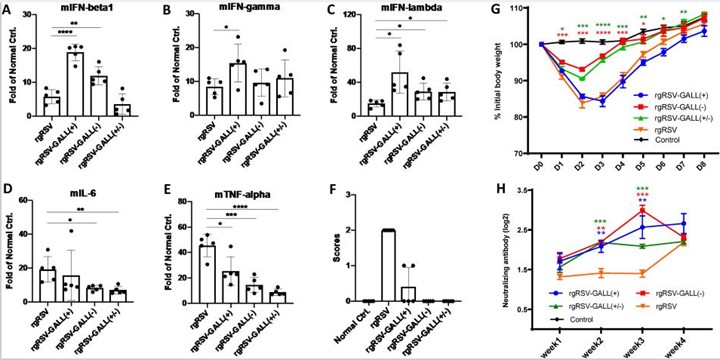
Innate immune response and antibody response of m6A-deficient rgRSVs in mice.
(A-E) Innate immune response of m6A-deficient rgRSVs in mice. 4-week old BALB/c mice were intranasally inoculated with PBS or PBS containing 5×105 TCID50 of rgRSV, rgRSV-GALL(+), rgRSV-GALL(-), or rgRSV-GALL(+/-). At 48h post-inoculation, all mice were euthanized, mouse IFNβ (mIFNβ) (A), mIFN-γ (B), mIFN-λ (C), mIL-6 (D), and mTNF-α (E) were quantified by real-time RT-PCR. (F) Clinical signs of mice inoculated with recombinant viruses. The severity of clinical signs were scored. Grade 3 (severe) was characterized by ruffled fur, hyperexcitability, tremors, and circling; grade 2 (moderate) was characterized by ruffled fur, hunched posture, and reduced food and water intake; grade 1 (mild) was characterized by mild ruffled fur; and grade 0 was defined as no symptoms. (G) Dynamics of mouse body weight after inoculation with m6A-deficient rgRSV. Five 4-6-week-old female BALB/c mice in each group were intranasally inoculated with PBS or 106 TCID50 of rgRSV-GALL(+), rgRSV-GALL(-), rgRSV-GALL(+/-), and parental rgRSV. The body weight for each mouse was evaluated at indicated time points. The average body weights of five mice are shown. (H), The RSV-neutralizing antibody titer was determined using a plaque reduction assay. Cytokine data are the fold change of mice inoculated rgRSV or each rgRSV mutant relative to the normal control mice inoculated with PBS. Data are the mean of 5 mice (n = 5) ± standard deviation. Antibody titers are the geometric mean titer (GMT) of 5 mice (n = 5) ± standard deviation. *P<0.5, **P < 0.01, ***P < 0.001, ****P < 0.0001. Data were analyzed using Student’s t-test.
ADVANTAGES
ADVANTAGES
-
Universal target for attenuation: m6A is a common modification in NNS RNA viruses, making it a broad target for vaccine development.
-
Grow to reasonably high titer: m6A-deficient viruses can be produced in high quantities, making vaccine production economically feasible.
-
Enhanced genetic stability: Reducing m6A modification sites requires multiple mutations, making it more difficult for the virus to revert to wild-type
-
Sufficient attenuation: m6A-deficient viruses show various degrees of attenuation, making them suitable for vaccine development.
-
Retain high immunogenicity: m6A-deficient viruses still induce strong immune responses without enhancing lung damage.
-
Trigger a higher innate immune response: m6A-deficient viruses may elicit stronger type I interferon responses, promoting better adaptive immune responses.
APPLICATIONS
-
Enhanced RSV vaccine development
-
Live vaccines for NNS viruses
-
Broad-spectrum antiviral drugs
PUBLICATIONS
-
Xue M, Zhang Y, Wang H, Kairis EL, Lu M, Ahmad S, Attia Z, Harder O, Zhang Z, Wei J, Chen P, Gao Y, Peeples ME, Sharma A, Boyaka P, He C, Hur S, Niewiesk S, Li J. Viral RNA N6-methyladenosine modification modulates both innate and adaptive immune responses of human respiratory syncytial virus. PLoS Pathog. 2021 Dec 20;17(12):e1010142. doi: 10.1371/journal.ppat.1010142. PMID: 34929018; PMCID: PMC8759664.
June 2, 2024
Proof of concept
Patent Issued
Licensing,Co-development
Chuan He